Most of the things in our “Treasures” series are living organisms. I think this is partly because lots of living organisms are easy to identify with: they exist on a scale similar to ours and are easy to categorize as discrete entities. Phenomena are a little harder to sell, for the most part. Stellar nucleosynthesis has had some help though, in the form of Carl Sagan’s wildly popular and surprisingly durable “star stuff” monologue. And it’s true: we’re literally made of atoms that came here from dying stars. Of course this is equally true of centipedes, norovirus and Rob Ford, so admittedly the magic relies on a bit of anthropocentrism.
But while the original is now nearly a quarter-century old, the basic process still holds more than enough wonder to power a brief voyage on the Ship of the Imagination™ (length of voyage largely determined by your choice of refreshments). The key, which is basically omitted from every version of the Sagan quote that I’ve heard, is that all the naturally occurring elements don’t just come from stars: they all represent stages in a single transformation that spans the entire life of a massive star.
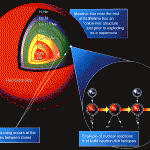
Stars generate energy from nuclear fusion. The stars’ immense gravity compresses hydrogen until the hydrogen atoms fuse together into helium—releasing vast quantities of energy in the process. This energy presses outward, preventing the star from collapsing under its own gravity. But as stars age, they eventually run out of hydrogen, leaving the star without the energy source that kept it from collapsing. At that point, the star’s core compresses under gravity, until the helium begins to fuse with the remaining hydrogen or more helium, producing lithium and beryllium. This then fuses with helium to produce…carbon. The building block of life was made from hydrogen deep in the hearts of ancient stars.
Near the end of its life, a star will fuse carbon and helium to form oxygen. Very large stars—those larger than eight times the size of our own sun—even have enough gravity to fuse carbon, producing neon, magnesium and other elements. In those stars, when the carbon has been exhausted, gravity may provide enough energy for neon and oxygen to fuse. This phase doesn’t last long; a star may fuse all its available oxygen in a year, leaving little besides silicon and sulfur. During the last 24 hours of its life, the star fuses its silicon, producing large quantities of nickel and iron and smaller quantities of some other elements. This is the bottom of the hill for “ordinary” stellar nucleosynthesis—and it’s the source of all the oxygen, carbon, iron, nickel, silicon and other common elements in the universe.
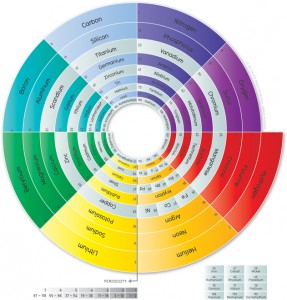
But what about other elements? Heavier elements come to be in the last few seconds of a large star’s fiery death, fused in the transformative fire of a supernova and blown outward, riding massive shockwaves throughout the universe. The heavy elements on this planet, and in our bodies, came not only from stars, but from the fiery death of unimaginable stellar explosions.
I think that this aspect—transformation—adds an additional layer of cool beyond just common origin. In one sense, stellar nucleosynthesis represents an important example of the ability of simple mechanisms to produce complex results. In this case, heat and gravity over time have been enough to produce all of the elements needed for life to develop on Earth. It’s a hidden premise in many minds that complexity can only come from more complexity. Stellar nucleosynthesis is an elegant and direct example of exactly the opposite.
Thanks to Markus Hammonds of the University of Tokyo for reviewing this piece for accuracy.